Pesticides >> Insecticide >> Fenvalerate
Fenvalerate
Fenvalerate 95%TC,
Fenvalerate 5%SC
Insecticide, acaricide, ixodicide
pyrethroid
NOMENCLATURE
Common name fenvalerate (BSI, E-ISO, (m) F-ISO, ESA)
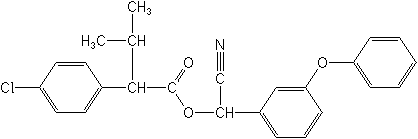
IUPAC name (RS)-a-cyano-3-phenoxybenzyl (RS)-2-(4-chlorophenyl)-3-methylbutyrate
Chemical Abstracts name cyano(3-phenoxyphenyl)methyl 4-chloro-a-(1-methylethyl)benzeneacetate
CAS RN [51630-58-1] unstated stereochemistry Development codes S-5602
(Sumitomo); WL 43 775 (Shell) Official codes OMS 2000
Fenvalerate APPLICATIONS
Mode of action Non-systemic insecticide and acaricide with contact
and stomach action. Uses Control of a wide range of pests, including
those resistant to organochlorine, organophosphorus, and carbamate
insecticides. Uses include control of chewing, sucking, and boring
insects (particularly Lepidoptera, Diptera, Orthoptera, Hemiptera,
and Coleoptera) in fruit, vines, olives, hops, nuts, vegetables,
cucurbits, cotton, oilseed rape, sunflowers, alfalfa, cereals, maize,
sorghum, potatoes, beet, peanuts, soya beans, tobacco, sugar cane,
ornamentals, forestry, and on non-crop land. Used for control of
flying and crawling insects in public health situations and in animal
houses. Also used as an animal ectoparasiticide. Formulation types
EC; UL; SC; WP. Compatibility Incompatible with alkaline materials.
Selected tradenames: 'Sumicidin' (Sumitomo); 'Arfen' (Ramcides);
'Cekufenvalerato' (Cequisa); 'Dufen' (Dhanuka); 'Fenkill' (United
Phosphorus); 'Fenny' (Nagarjuna Agrichem); 'Fenval' (RPG); 'Kayvalerate'
(Krishi Rasayan); 'Newfen' (Gharda); 'Sanvalerate' (Sanachem); 'Shasicidin'
(Sanonda); 'Starfen' (Shaw Wallace); 'Sumitox' (Aimco)
Fenvalerate OTHER TRADENAMES
'Bhasma' (Biostadt); 'Fencur' (BEC); 'Fenero' (Sabero); 'Fenirate'
(Agrochem); 'Krifen' (Krishi Rasayan); 'Parryfen' (Parry); 'Qingwujuzhi'
(Shenzhen Jiangshan); 'Shamiethrin' (Zhong-Xi); 'Stalker' (Indofil);
'Tribute' (Aventis); 'Triumphcard' (Dhanuka); 'Valour' (Crop Health);
'Vapcocidin' (Vapco); 'Vifenva' (Vipesco) mixtures: 'Fenikill' (+
fenitrothion) (Vapco); 'Miejingthrin' (+ propargite) (Zhong-Xi);
'Pre Ling' (+ phoxim) (Zhong-Xi); 'Vegehon' (+ dimethoate) (Agros);
'Vifensu' (+ fenitrothion) (Vipesco) Discontinued names: 'Belmark'
* (Cyanamid); 'Pydrin' * (Du Pont)
ANALYSIS
Product analysis of pyrethroids reviewed by E. Papadopoulou-Mourkidou
in Comp. Analyt. Profiles. By hplc (idem, ibid.; E. Papadopoulou-Mourkidou,
Anal. Methods Pestic. Plant Growth Regul., 1988, 16, 31; 1984, 13,
121) or by glc (CIPAC Handbook, 1988, D, 100; ibid., 1995, G, 68-70).
Residues determined by glc with ECD (ibid.; A. Ambrus et al., J.
Assoc. Off. Anal. Chem., 1981, 64, 733; Man. Pestic. Residue Anal.,
1987, I, 6, S19; Anal. Methods Residues Pestic., 1988, Part I, M11).
Details available from Sumitomo Chemical Co.
MAMMALIAN TOXICOLOGY
Reviews FAO/WHO 47 (see part 2 of the Bibliography). See also A.
J. Gray & D. M. Soderlund, Chapt. 5 in "Insecticides".
IARC ref. 53 class 3 Oral Acute oral LD50 for rats 451 mg/kg. Skin
and eye Acute percutaneous LD50 for rabbits 1000-3200, rats >5000
mg/kg. Slightly irritating to skin and eyes (rabbits). Inhalation
LC50 for rats >101 mg/m3. NOEL (2 y) for rats 250 mg/kg diet.
ADI (JMPR) 0.02 mg/kg b.w. [1986]. Toxicity class WHO (a.i.) II;
EPA (formulation) II
ECOTOXICOLOGY
Birds Acute oral LD50 for domestic fowl >1600, mallard ducks
9932 mg/kg. Dietary LC50 for quail >10 000, mallard ducks 5500
mg/kg. Fish LC50 (96 h) for rainbow trout 0.0036 mg/l. Bees Toxic
to bees. Contact LD50 0.23 mg/bee.
ENVIRONMENTAL FATE
EHC 95 (WHO, 1990). EHC 95 cites rapid degradation and decomposition,
reduced toxicity of its degradation products, and absence of leaching
in soil. Although laboratory tests show high toxicity for fish and
bees, these effects are markedly reduced under field conditions
because of adsorption to sediments, and the compound's strong repellent
effect. The report concludes that risks to the environment are unlikely
when it is applied as recommended. Animals In mammals, following
oral administration, fenvalerate is rapidly metabolised. Up to 96%
is excreted in the faeces within 6-14 days. For a study of the metabolism
of fenvalerate in the laying hen, see J. Agric. Food Chem., 1989,
37, 190. Plants In plants, fenvalerate is split into two parts by
cleavage of the ether group, followed by further hydroxylation in
the 2- and 4- positions of the phenoxy ring, and hydrolysis of the
nitrile group to amide and carboxyl groups. The majority of the
acids and phenols thus formed are converted into glucosides. Soil/Environment
In aqueous media, the ester bond is hydrolysed. In light, decarboxylation
occurs, with recombination of the cleaved moieties (R. L. Holmstead
et al., J. Agric. Food Chem., 1977, 25, 56-58). DT50 in soil c.
75-80 d.