Pesticides >> Insecticides>> Fenbutatin Oxide
Fenbutatin Oxide 55%SC
Acaricide
FRAC X
NOMENCLATURE
Common name Fenbutatin Oxide (BSI, E-ISO, JMAF); fenbutatin-oxyde
((m) F-ISO)
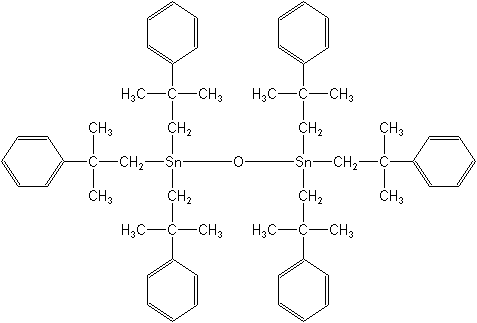
IUPAC name bis[tris(2-methyl-2-phenylpropyl)tin] oxide
Chemical Abstracts name hexakis(2-methyl-2-phenylpropyl)distannoxane
Other names hexakis CAS RN [13356-08-6] EEC no. 236-407-7 Development
codes SD 14 114 (Shell) Official codes ENT 27 738
Fenbutatin Oxide APPLICATIONS
Mode of action Non-systemic acaricide with contact and stomach action.
Uses Control of all motile stages of a wide range of phytophagous
mites on pome fruit, stone fruit, citrus fruit, soft fruit, vines,
bananas, cucurbits, ornamentals, and glasshouse crops. Gives long
residual control. Phytotoxicity Phytotoxic to tangerines, tangelos,
and some varieties of grapefruit. Otherwise, non-phytotoxic when
used as directed. Formulation types SC; WP. Selected tradenames:
'Osadan' (Japan only) (BASF); 'Torque' (BASF); 'Vendex' (Du Pont,
BASF)
OTHER TRADENAMES
'Torq' (Fargro) mixtures: 'Mitedown' (+ polynactins) (Eikou Kasei)
ANALYSIS
Product analysis by potentiometric non-aqueous titrimetry (CIPAC
Handbook, 1988, D, 77). Residues determined by glc with ECD or FPD
of suitable derivative (Anal. Methods Pestic. Plant Growth Regul.,
1978, 10, 139; Methodensammlung Rückstandsanal. Pflanzenschutzmitteln,
1987, S24; M. Sano et al., J. Assoc. Off. Anal. Chem., 1979. 62,
764). Details available from BASF.
MAMMALIAN TOXICOLOGY
Reviews FAO/WHO 65, 67 (see part 2 of the Bibliography). Oral Acute
oral LD50 for rats 2631, mice 1450, dogs >1500 mg/kg. Skin and
eye Acute percutaneous LD50 for rabbits >2000, rats >1000
mg/kg. Irritating to skin and severely irritating to eyes. Inhalation
LC50 0.23 mg a.i. (as dust)/l. NOEL (2 y) for rats 100 mg/kg diet,
for dogs 15 mg/kg daily. ADI (JMPR) 0.03 mg/kg b.w. [1992]. Toxicity
class WHO (a.i.) III (Table 5); EPA (formulation) III EC hazard
T+; R26| Xi; R36/38| N; R50, R53
ECOTOXICOLOGY
Birds Dietary LC50 (8 d) for bobwhite quail 5065 mg/kg diet. Fish
LC50 (48 h) for rainbow trout 0.27 mg a.i./l (as WP formulation).
Daphnia LC50 (24 h) 0.05-0.08 mg/l. Bees Acute oral LD50 >0.1
mg/bee. Other beneficial spp. No adverse effects to a wide range
of predatory and parasitic arthropods.
Fenbutatin Oxide: ENVIRONMENTAL FATE
Soil/Environment In soil, it is metabolised to dihydroxy-bis(2-methyl-2-phenylpropyl)stannane
and 2-methyl-2-phenylpropyl stannonic acid, presumably ultimately
forming tin oxide/hydroxide. In tests in commercial use, there was
minimal movement of Fenbutatin Oxide or its metabolites out of the
top 30 cm of soil (A. Gray et al., Pestic. Sci., 43, 295-302 (1995))