Pesticides >> Insecticides >> Chlorpyrifos
Chlorpyrifos
Chlorpyrifos 95%TC
Chlorpyrifos 48%EC
Insecticide
IRAC 1B; organophosphate
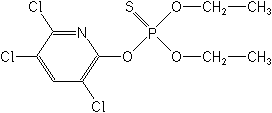
Chlorpyrifos NOMENCLATURE
Common name chlorpyrifos (BSI, E-ISO, ANSI, ESA, BAN); chlorpyriphos ((m) F-ISO, JMAF); chlorpyriphos-éthyl ((m) France)
IUPAC name O,O-diethyl O-3,5,6-trichloro-2-pyridyl phosphorothioate
Chemical Abstracts name O,O-diethyl O-(3,5,6-trichloro-2-pyridinyl) phosphorothioate
CAS RN [2921-88-2] EEC no. 220-864-4 Development codes Dowco 179 (Dow) Official codes OMS 971; ENT 27 311
Chlorpyrifos PHYSICAL CHEMISTRY
Mol. wt. 350.6 M.f. C9H11Cl3NO3PS Form Colourless crystals, with a mild mercaptan odour. M.p. 42-43.5 ºC V.p. 2.7 mPa (25 ºC) KOW logP = 4.7 Henry 6.76 10-1 Pa m3 mol-1 (calc.) S.g./density 1.44 (20 °C) Solubility In water c. 1.4 mg/l (25 ºC). In benzene 7900, acetone 6500, chloroform 6300, carbon disulfide 5900, diethyl ether 5100, xylene 5000, iso-octanol 790, methanol 450 (all in g/kg, 25 ºC). Stability Rate of hydrolysis increases with pH, and in the presence of copper and possibly of other metals that can form chelates; DT50 1.5 d (water, pH 8, 25 ºC) to 100 d (phosphate buffer, pH 7, 15 ºC).
Chlorpyrifos COMMERCIALISATION
History Insecticide reported by E. E. Kenaga et al. (J. Econ. Entomol., 1965, 58, 1043). Commercially introduced by Dow Chemical Co. (now Dow AgroSciences) during 1965. Patents US 3244586 Manufacturers Agriphar; Agrochem; Aimco; Cheminova; Crystal; DE-NOCIL; Dow AgroSciences; Drexel; Excel; Ficom; Gharda; Hesenta; Luxembourg; Makhteshim-Agan; Mitsu; Nagarjuna Agrichem; Rallis; Sharda; Sinon; Sundat; Tagros
Chlorpyrifos APPLICATIONS
Biochemistry Cholinesterase inhibitor. Mode of action Non-systemic insecticide with contact, stomach, and respiratory action. Uses Control of Coleoptera, Diptera, Homoptera and Lepidoptera in soil or on foliage in over 100 crops, including pome fruit, stone fruit, citrus fruit, nut crops, strawberries, figs, bananas, vines, vegetables, potatoes, beet, tobacco, soya beans, sunflowers, sweet potatoes, peanuts, rice, cotton, alfalfa, cereals, maize, sorghum, asparagus, glasshouse and outdoor ornamentals, turf, and in forestry. Also used for control of household pests (Blattellidae, Muscidae, Isoptera), mosquitoes (larvae and adults) and in animal houses. Phytotoxicity Non-phytotoxic to most plant species when used as recommended. Poinsettias, azaleas, camellias, and roses may be injured.