Products >> Fungicides >> Cymoxanil
Cymoxanil
Cymoxanil 98%TC
Cymoxanil 70%WG
Cymoxanil 45%WG
Cymoxanil 30%WG
Fungicide
FRAC X
Cymoxanil NOMENCLATURE
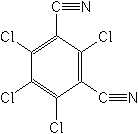
Common name cymoxanil (BSI, ANSI, draft E-ISO, (m) draft F-ISO)
IUPAC name 1-(2-cyano-2-methoxyiminoacetyl)-3-ethylurea
Chemical Abstracts name 2-cyano-N-[(ethylamino)carbonyl]-2-(methoxyimino)acetamide
CAS RN [57966-95-7] EEC no. 261-043-0 Development codes DPX-T3217
(Du Pont)
Cymoxanil APPLICATIONS
Mode of action Foliar fungicide with protective and curative action.
Has contact and local systemic activity, and also inhibits sporulation.
Uses Control of Peronosporales, especially Peronospora, Phytophthora,
and Plasmopara spp. Normally used in combination with protectant
fungicides (to improve residual activity) on a range of crops, including
vines, hops, potatoes, and tomatoes. Formulation types WP. Compatibility
Incompatible with alkaline materials. Selected tradenames: 'Curzate'
(Du Pont); 'Asco' (Agrimix); mixtures: 'Aktuan' (+ dithianon) (BASF);
'Fobeci' (+ benalaxyl+ folpet) (Sipcam Inagra); 'Manex C-8' (+ mancozeb)
(Griffin); 'Micexanil' (+ mancozeb) (Sipcam); 'Pulsan' (+ mancozeb+
oxadixyl) (Syngenta); 'Quadris' (+ azoxystrobin) (Syngenta); 'Sygan
PM' (+ folpet+ mancozeb) (Griffin); 'Syphal PM' (+ copper oxychloride+
folpet+ mancozeb) (Griffin)
Cymoxanil OTHER TRADENAMES
'Cimoxpron' (Probelte); 'Sarman' (Sipcam Phyteurop); 'Texas' (Rocca);
'Vitene' (Sipcam); 'Zetanil' (Sipcam) mixtures: 'Copral' (+ copper
sulfate) (Du Pont); 'Equation Pro' (+ famoxadone) (Du Pont); 'Sirdate
S' (+ folpet+ oxadixyl) (Du Pont); 'Sygan LS' (+ folpet+ mancozeb)
(Du Pont); 'Sygan S' (+ folpet) (Du Pont); 'Syphal LS' (+ copper
oxychloride+ folpet+ mancozeb) (Du Pont); 'Apron Elite' (+ carbendazim+
oxadixyl+ thiram) (Syngenta); 'Ashlade Solace' (+ mancozeb) (Ashlade);
'Aviso Cup' (+ copper oxychloride+ metiram) (BASF); 'Curtine-V'
(+ mancozeb) (Vapco); 'Cymotine' (+ mancozeb) (Agro Chemicals);
'Eclair' (+ trifloxystrobin) (Bayer); 'Horizon' (+ famoxadone) (Du
Pont, Nissan); 'Kupfer Fusilan' (+ copper oxychloride) (Kwizda);
'Manoxil' (+ mancozeb) (AgroSan); 'Ripost Pepite' (+ mancozeb+ oxadixyl)
(Syngenta); 'Sirdate P' (+ maneb+ oxadixyl) (Griffin); 'Valiant'
(+ folpet+ fosetyl-aluminium) (Aventis); 'WAKIL XL' (+ fludioxonil+
metalaxyl-M) (Syngenta) Discontinued names mixtures: 'Besiege WSB'
* (+ mancozeb) (Du Pont); 'Systol M' * (+ mancozeb) (Du Pont); 'Trustan'
* (+ mancozeb+ oxadixyl) (Du Pont)
Cymoxanil ANALYSIS
Product analysis by hplc. Residues determined by glc. Details available
from Du Pont.
MAMMALIAN TOXICOLOGY
Oral Acute oral LD50 for male and female rats 960 mg/kg. Skin and
eye Acute percutaneous LD50 for rabbits >2000 mg/kg. Not an eye
irritant; slight skin irritant (rabbits). Not a skin sensitiser
(guinea pigs). Inhalation LC50 (4 h) for male and female rats >5.06
mg/l. NOEL (2 y) for male rats 4.1, female rats 5.4, male mice 4.2,
female mice 5.8, male dogs 3.0, female dogs 1.6 mg/kg b.w. daily.
ADI 0.016 mg/kg. Toxicity class WHO (a.i.) III; EPA (formulation)
III EC hazard Xn; R22| R43| N; R50, R53
Cymoxanil ECOTOXICOLOGY
Birds Acute oral LD50 for bobwhite quail and mallard ducks >2250
mg/kg. Dietary LC50 (8 d) for bobwhite quail and mallard ducks >5620
mg/kg diet. Fish LC50 (96 h) for rainbow trout 61, bluegill sunfish
29, common carp 91, sheepshead minnow >47.5 mg/l. Daphnia LC50
(48 h) 27 mg/l. Algae 23% Inhibition of Selenastrum capricornutum
at 1.05 mg/l after 5 d; EC50 for Anabaena flos-aquae 231 ppb. Other
aquatic spp. LC50 (96 h) for Eastern oyster >46.9, mysid shrimp
>44.4 ppm. Bees Not toxic to bees; LD50 (48 h, contact) >25
mg/bee; LC50 (48 h, oral) >1000 ppm. Worms LC50 (14 d) >2208
mg/kg soil.
Cymoxanil ENVIRONMENTAL FATE
Animals In rats, cymoxanil was readily absorbed through the intestines;
the majority of the dose was excreted in the urine. The proposed
metabolic pathway involves hydrolysis of cymoxanil and then degradation
to glycine. Plants Degraded to glycine with subsequent incorporation
into natural products (proteins and starch). Soil/Environment In
lab. soils, DT50 0.75-1.5 d (5 soils, pH range 5.7-7.8, o.m. 0.8-3.5%).
In the field, DT50 (bare soil) 0.9-9 d. In aquatic studies, DT50
<1 d. Koc 39-250. Cymoxanil is mobile, adsorption Freundlich
K 0.29 to 2.86 in four soil types.